METHANE HYDRATES
Methane trapped in marine sediments as a hydrate represents such an immense carbon reservoir that it must be considered a dominant factor in estimating unconventional energy resources; the role of methane as a 'greenhouse' gas also must be carefully assessed. Dr. William Dillon, U.S. Geological Survey

From the USGS website:
- Hydrates store immense amounts of methane, with major implications for energy resources and climate, but the natural controls on hydrates and their impacts on the environment are very poorly understood.
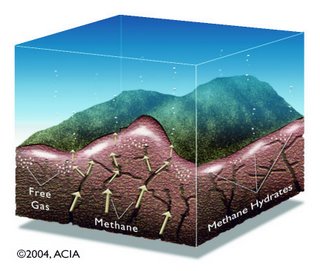
- Gas hydrates occur abundantly in nature, both in Arctic regions and in marine sediments. Gas hydrate is a crystalline solid consisting of gas molecules, usually methane, each surrounded by a cage of water molecules. It looks very much like water ice. Methane hydrate is stable in ocean floor sediments at water depths greater than 300 meters, and where it occurs, it is known to cement loose sediments in a surface layer several hundred meters thick.
- The worldwide amounts of carbon bound in gas hydrates is conservatively estimated to total twice the amount of carbon to be found in all known fossil fuels on Earth.
Now, the news:
Undersea Gas Could Speed Global Warming - Study
July 21, 2006 (Reuters)
- If the world continues to get warmer, vast amounts of methane gas trapped in ice under the sea could belch up and worsen climate change, according to a study.
- "We may have less time than we think to do something (about the prospect of global warming)," Dr. Ira Leifer, a marine scientist at University of California Santa Barbara, said in an interview.
- Leifer is the main author of a study that looks at how "peak blowouts" of melting undersea formations called methane hydrates could release the potent greenhouse gas into the atmosphere. The study was published Thursday in Global Biogeochemical Cycles, a climate science publication.








2 Comments:
Here are some links that I believe will be interested
I say briefly: Best! Useful information. Good job guys.
»
Post a Comment
<< Home